Anything that has mass and occupies space is called matter. Matter is everything around you. Atoms and compounds are all made of very small parts of matter. Those atoms go on to build the things you see and touch every day. Matter is defined as anything that has mass and takes up space (it has volume).
Composition of matter:
Chemistry is used all around us. The matter is composed of different building blocks.
- Atom – Matter is made up of tiny building blocks called atoms. It takes millions and millions of atoms to make even the smallest object.
- Element – Substances made up of a single type of atom are called elements.
- Compound – Compounds are made up of different types of elements.
- Mixture – A material made up of different types of compounds that do not chemically combine. A molecule can be made of the same kind of atoms. Example: Oxygen, hydrogen. Molecules can be made of different kinds of atoms. Example: Carbon dioxide is made of carbon and oxygen, ammonia is made of nitrogen and hydrogen.
Characteristics of matter:
Molecules of the matter show constant motion because of kinetic energy. All particles of matter have the following characteristics:
- Particles of matter are very small in size(cannot see them). Example- one drop of water contains approx 1021 particles of water.
- Molecules of matter are held together by intermolecular forces of attraction. Spaces exist between molecules of matter. These gaps are called intermolecular spaces.
- Particles of matter are in continuous motion(this zig zag motion of the particles is Brownian motion).
- Particles of matter attract one another.This is because of some force of attraction which is called Intermolecular force of attraction that keep them together.
States of matter:
Depending on the arrangement of particles, their intermolecular space and intermolecular force of attraction, we can find three states of matter:
- Solid
- Liquid
- Gas.
SOLIDS
Molecules in solids are packed tightly together, they have strong forces of attraction between them and very small or negligible gaps between them. They are not free to move and can only vibrate about their mean positions. So, they have fixed shape and size and are hard and difficult to compress.
Properties of solids:
- Solids have definite volume.
- Solids have definite shape.
- Solids have definite shape.
- Solids are rigid (non-bending or inflexible) and retain their shape.
- Solids do not flow.
- Solids are in-compressible.
- Solids have high density.
- Solids can have any number of free surface.
- Solids exert pressure at their base due to their weight.
- Solids show low thermal expansion.
- Solids do not diffuse easily into other solids.
LIQUIDS
Molecules in liquids are packed less closely than solids. They have lesser forces of attraction and more spaces between them. This allows the molecules to move more freely. Thus, a liquid can flow and take the shape of the container into which they are poured. So, they have fixed volume but no definite shape.
Properties of liquids:
- Liquids have a definite volume and shape.
- Liquid are not rigid.
- Liquids can flow easily
- Liquids are slightly compressible.
- Liquids have low density in comparison to solids.
- Liquids have only one free surface.
- Liquids exert pressure in all directions.
- Liquids show high thermal expansion.
- Liquids can easily diffuse into other liquids.
- They tends to acquire minimum surface area. So, they have a tendency to form a drop.
GASES
Molecules of gas have large spaces between them and very little force of attraction. They occur far apart from each other and have great freedom of movement. They have neither fixed shape nor fixed volume. They can expand to fill a bigger container and can be compressed to fit into a small container.
Properties of gases:
- Gases neither have a definite volume and nor a definite shape.
- Gases are not rigid.
- Gases flow in all directions.
- Gases are highly compressible.
- Gases have very low density.
- They do not have any free surface.
- They exert pressure from all direction on the walls of the container in which they are kept.
- They show very high thermal expansion .
- It can diffuse into other gases very fast.
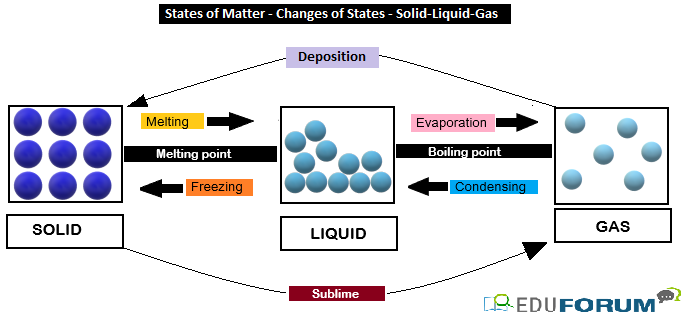
Difference in States of Matter 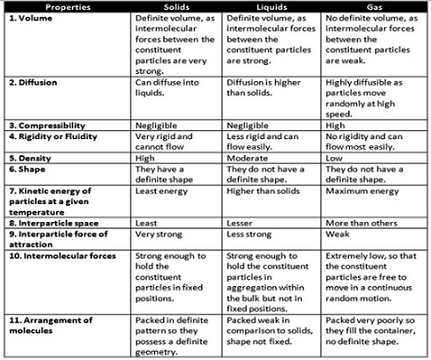 Changes in state of matter:
Changes in state of matter:
The physical state of matter can be changed by heating or cooling. The change of matter from one physical state to another is called change of state.
- Melting: Change of a solid into a liquid when heat is applied. In a pure crystalline solid, this process occurs at a fixed temperature called the melting point; an impure solid generally melts over a range of temperatures below the melting point of the principal component.
- Boiling: The process during which a liquid changes into its gaseous state at a fixed temperature on heating is called boiling.
- Evaporation: The process during which a liquid changes into its gaseous at any temperature below boiling point is called evaporation.
- Sublimation: The process during which a solid directly changes into its gaseous state on heating is called sublimation.
- Deposition: The process during which a gas directly changes into its solid state by cooling is called deposition.